Pulmonary embolism is formed when a portion of a blood clot breaks off and travels through the blood stream, typically the deep venous system, passes through the right side of the heart (right atrium and right ventricle), and becomes lodged in a pulmonary artery. This can cause partial or complete obstruction of the involved pulmonary artery. Such an obstruction impedes blood flow to the affected portion of the lung resulting in dead space. The affected portion of the lung does not participate in its primary function of oxygenating the blood and removing carbon dioxide from the de-oxygenated blood. Poor oxygenation results in poor delivery of oxygen to the vital organs which may subsequently malfunction or fail to function. A secondary effect is that with obstruction of a pulmonary artery, there is an increase in pulmonary arterial pressure, right heart pressure, and resultant right heart strain. This can result in right-sided heart failure, ischemia, and death. Delay of treatment or lack of treatment results in increased morbidity and mortality [1].
Pulmonary emboli affect between 300,000-600,000 Americans every year and result in 12,000-80,000 deaths/year in the United States [2]. Accurate numbers for Europe are scarce. However, the incidence of PE has been reported to be 6-20.8 cases/10,000 people/year in Brittany, France and Malmö, Sweden respectively [3]. Extrapolating those statistics, there would be an estimated 19,000 cases of PE per year in Sweden and 39,480 cases of PE per year in France. According to the ministry of health in Catalonia, there are an estimated 60,000 cases of PE a year in Spain [4].
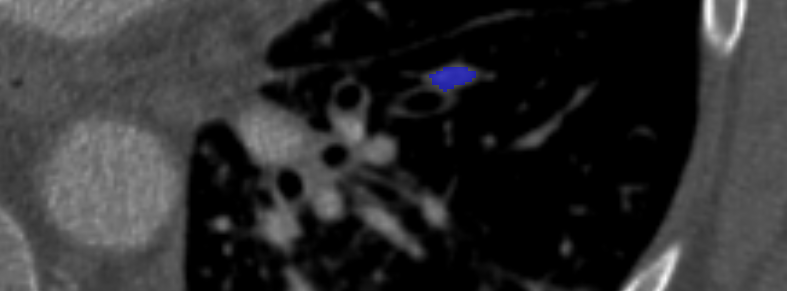
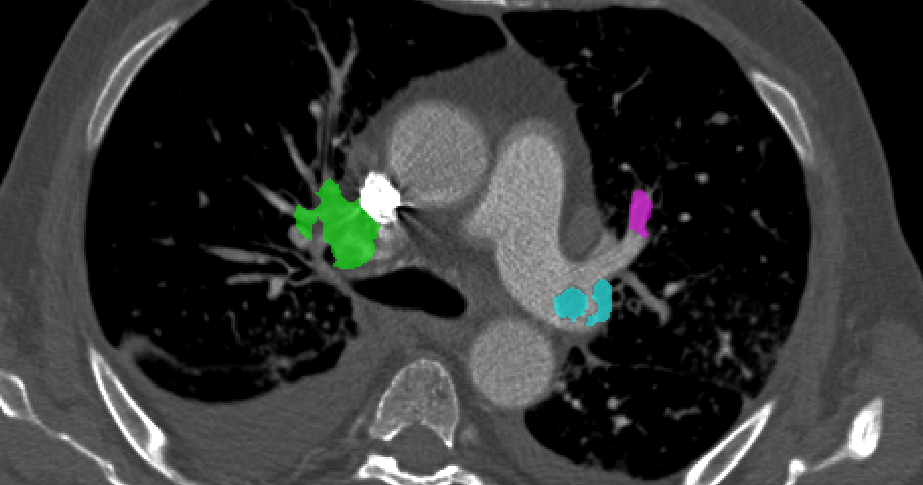
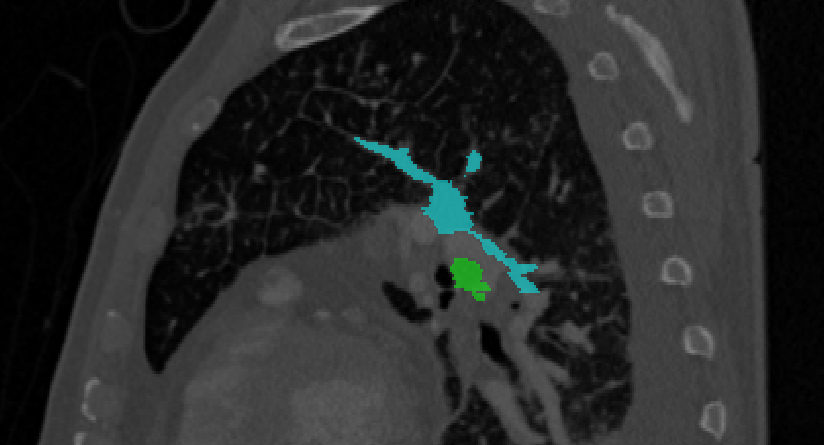
The clinical presentation of PE is variable. Common symptoms include shortness of breath, chest pain, and rapid heart beat (tachycardia). Additional symptoms may include coughing up blood (hemoptysis), low blood pressure (hypotension) and loss of consciousness. Unfortunately, these symptoms are nonspecific and the clinical diagnosis can be challenging. Computed tomography pulmonary angiography (CTPA) is now accepted as the diagnostic imaging study of choice to confirm the clinical suspicion of acute PE. Because the clinical and laboratory diagnosis of PE are challenging, only a small fraction of all patients who undergo CTPA have PE. The diagnosis of PE by CTPA is made by the identification of a filling defect in a pulmonary artery.
Based on CTPA interpretation with respect to an expert reference standard, radiologists’ sensitivity ranges from 77-94% [5, 6]. Computer aided detection (CAD) systems have been developed to augment radiologists’ sensitivity [7, 8, 9, 10, 11, 12]. Data suggests that radiologists who use current CAD systems can improve the sensitivity to 92-98% [5, 6]. However, the drawbacks of current systems preclude clinical use, and few if any, radiologists use CAD to assist in the interpretation of CTPA. The greatest drawback of current CAD systems is the high false positive rate (4.9-19 False positives/study). To date, there is no data on the clinical impact (e.g. mortality from PE after CTPA) of an improved sensitivity for the detection of PE. However, one can assume that the delay of treatment or lack of treatment from PE detected by CAD that would otherwise remain non-detected will improve patient care. Given the clinical relevance of pulmonary embolism, we have joined efforts to produce an annotated database of CTPA scans where all the emboli are detected and segmented. An example of the scan is shown Fig. 1. Such scans and reference standard are proposed as a benchmark for testing different CAD algorithms for PE. We challenge the scientific community to develop and test their algorithms with this dataset.